Translations may contain older or less detailed information than the original German page!
Normal histamine metabolism in healthy people
Page overview:
Summary:
Properties of histamine:
Histamin is a naturally occurring biogenic amine. This small molecule is well soluble in water but poorly soluble in fat. It is stable in heat and cold and can not be removed from food neither by boiling nor by freezing.
Biosynthesis:
In living organisms, histamine is synthesized from histidine (which is an amino acid) by an enzyme named histidine-decarboxylase (HDC).
Occurrence:
In animals, histamine has many important biological functions. It is also common in bacteria and plants. Animals produce is in certain specialized blood-/tissue-/immune cells (especially in mastocytes and basophils). It is stored in hundreds of little "bubbles" inside mast cells (mastocytes). When a mast cell detects an "enemy" or a threat (microorganism, substances or other triggers), this stored histamine and other mediators can be released immediately in order to alert the surrounding tissues by these chemical signals (mediators). After release, it is circulating between cells and in the blood.
Main triggers are immune reactions against diseases and allergens. Additionally, also physical, chemical and psychological stimuli can lead to sudden or slow histamine release. Chemical stimuli are called liberators.
Histamine appears in spoilt food and in microbial fermentation and ripening and is also produced by the intestinal flora. Diamine oxidase (DAO) acts as an enzymatical barrier. It decomposes histamine in the intestine and prevents the body from histamine poisoning.
Functions of histamine:
Histamine is regulating many metabolic processes. Its most important function is probably to alert the body in case of microbial attacks.
Signalling pathway: Histamine activates four different known histamine receptors (H1, H2, H3, H4) located on the surface of the cells. They are transmitting the chemical signal into the cell. Furthermore, histamine can also enter the cell by active transmembranal transport mechanisms. Signal proteins can be activated by monoaminylation, which causes long term hormonal reactions, but is still poorly understood.
H1-receptors are transmitting the most important effects of histamine: Allergy-like symptoms like itching and pain, inflammation, muscle contraction in bronchi and large blood vessels and enlargement of small blood vessels, blood pressure, regulation of body temperature.
H2-receptors are enhancing the production of gastric acid, they are accelerating the intestinal motility (diarrhea) and the heartbeat.
H3-receptors are inhibiting further release of histamine. Release of other mediators is regulated.
H4-receptors are directing immune cells towards the source of histamine, into inflammation hotspots.
Decomposition (degradation):
Diamine oxidase (DAO) decomposes histamine, especially in the intestine before uptake.
Histamine-N-methyltransferase (HNMT) is able to decompose histamine inside the cells.
Furthermore, monoaminoxidase B (MAO-B) is able to degrade histamine.
Chemical characteristics of histamine
Histamine (combining the Greek "histos" = tissue and "amin" = nitrogenous compound) is a natural substance, which fulfils important regulatory and mediatory functions in animal organisms, and is also frequently found in bacteria and in the plant kingdoms. Biochemically, histamine belongs to the group of biogenic amines. In its purest form, histamine is a white solid material with a molar mass of 111.15 g/mol. Histamine is easily soluble in water and ethanol, but badly dissolved in diethyl ether, which indicates that it has also bad solubility in fat.
Molecular formula: C5H9N3
Structural formula: 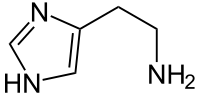
Synonyms: 2-(1H-imidazol-4-yl)ethanamine
CAS number: 51-45-6
Functions of histamine
In the human organism, histamine fulfils various functions as tissue hormone, neurotransmitter and messenger substance (biochemical signal transduction), which have only been insufficiently studied so far. At a molecular level, histamine exerts its actions via an activation of histamine receptors H1, H2, H3 and H4, which are part of the family of G protein-coupled receptors (GPCR). Histamine acts by binding to target receptors according to the lock and key model and modulating intracellular signal cascades (signal transduction chains).
An activation of H1 receptors is primarily responsible for the allergy symptoms triggered by histamine. These include itching and pain, contractions of smooth muscle tissue in the bronchial tubes and large blood vessels (diameter of more than 80 µm) as well as dilation of smaller blood vessels with hives and flush. In the central nervous system, histamine is involved in triggering vomiting and the regulation of the sleep-wake cycle via an activation of H1 receptors. H1 receptors also play a part in regulating the release of hormones such as adrenaline. Histamine is a messenger substance active in inflammatory processes and burns, and furthermore boosts the release of additional inflammatory mediators. Besides, it seems to play a part in the regulation of body temperature, the central control of blood pressure and pain perception. [Wikipedia: Histamine]
H2 receptors are involved in the regulation of gastric acid production and bowel movements (motility, peristalsis). An increase of gastric acid production may be interpreted as a component of histamine-induced immune reaction. An accelerated onward transport of the intestinal contents leads to diarrhea and may also be seen as an immune response. A stimulation of H2 receptors also leads to an accelerated or stronger heart beat as well as dilation of smaller blood vessels.
In the human body the H3 receptors are primarily found presynaptically on cells of the central and peripheral nervous systems. As autoreceptors they play a role when negative feedback prevents additional histamine release. Via presynaptic receptors (in particular H2 receptors), histamine has a regulatory influence on noradrenergic, serotoninergic, cholinergic, dopaminergic and glutaminergic neurons by blocking the release of neurotransmitters in the central and peripheral nervous systems. Thus it inhibits the release of the neurotransmitters acetylcholine, noradrenalin and serotonin as heteroreceptor. In this way, histamine influences indirectly the activity of these neurotransmitters. Through these mechanisms, the H3 receptors play a role in the central regulation of hunger and thirst, the circadian rhythm, body temperature and blood pressure. Furthermore, these receptors are said to be directly or indirectly implicated in the pathophysiology of neurological pain, schizophrenia, Parkinson's disease and ADHS.
H4 receptors are involved in the targeted migration of immune cells such as eosinophil granulocytes, T lymphocytes and monocytes to sources of histamine. This is why it is assumed that these receptors play an important role in the recruitment of leukocytes during immune responses, in particular in allergic reactions.
Monoamine transporters regulate cytological processes
Unlike previously believed, water-soluble hormones such as serotonin, histamine and catecholamine do not only act via receptors on the cell's surface but also through monoamine transporters (MATs) within the cells. Similar to phosphorylation, monoamine transporters have profound effects on various cytological processes, which still need further clarification [Vowinckel 2012; Walther; Stahlberg and Vowinckel 2011; Walther 2007; Scinexx 2009]. With antihistamines - which are only really ever specific to a single type of receptor - such cytological modifications cannot be prevented.
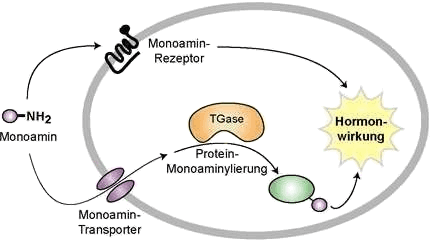
Histamine and other water-soluble monoamines cannot pass the cell membrane by diffusion. A considerable part of their effects are triggered by numerous membranous monoamine receptors (histamine receptors). Specific monoamine transporters, however, accumulate histamine and other hormones in the cytoplasm, where the monoamines constitutively activate signal proteins through transglutaminase (TGase) by monoamine transporters. This leads to longer lasting hormone activity similar to those observed in thrombocytes and in muscle cells of blood vessels. [Walther 2007]
Biosynthesis of histamine
Histamine is derived from the decarboxylation of the amino acid histidine, a reaction catalyzed by the enzyme L-histidine decarboxylase.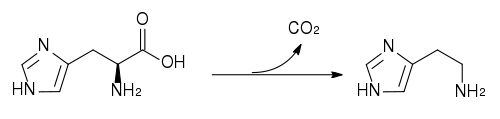
The ability to synthesize histamine is very common in microorganisms such as bacteria or yeast, as well as in the animal and plant kingdoms.
Occurrence of histamine
Endogenous histamine (=produced naturally in the body)
Storage
Histamine is a substance produced by the organism itself and stored in blood and tissue cells. The storage takes place by binding to heparin in so-called vesicles. These are small organelles, separated by membranes, inside the cell. Enclosed in these vesicles, histamine is immobilized and cannot do any harm, but it is always able to be immediately released if required. Histamine is particularly synthesized and stored in the following cell types:
- Mast cells [Jarisch 2004]
- Basophile granulocytes [Jarisch 2004]
- Neurons and other nerve cells, such as cerebrovascular endothelial cells [Hough 1999]
The basophile granulocytes are blood cells. Mast cells are found in tissue, in particular in epidermal cells of the skin, in histamine-storing cells of the mucous membranes, in bronchial tubes, the gastro-intestinal system (i.e. gastric mucosa) and the brain. The highest histamine concentrations can be measured in the hypothalamus [Jarisch 2004; Hough 1999; Wikipedia: Histamine].
Release of histamine
Histamine is released from the vesicles during IgE-induced immediate-type (type I) allergic reactions or through complement factors such as for example an endotoxin-induced shock. In addition to tissue hormones, medications such as opiates, muscle relaxants, plasma expanders and radiocontrast agents, may trigger the release of histamine (please refer to Liste unverträglicher Medikamente). Other important storage sites of histamine are the ECL cells of the gastric mucosa, which can release histamine induced by hormones and tissue hormones such as gastrin, acetylcholine and pituitary adenylate cyclase activating polypeptide (PACAP).
A release of histamine in the synaptic cleft of histaminergic neurons is inhibited by acetylcholine, noradrenalin, and histamine via the presynaptic receptors.
Exogenous histamine (ingested by food)
For histamine intolerance, exogenous (=coming from the outside) histamine is probably even more relevant than histamine produced by the body itself. Histamine is a component that can be found in most foods in varying concentrations. It occurs when food perishes, particularly products that are fermented, matured or have been stored over a long period of time. An additional, but less important source of exogenous histamine is the gut flora. Many kinds of microorganisms found in the intestines are able to produce histamine.
Histamine ingested with food or produced in the gut must not get into the body, because an increased histamine level (histamine poisoning) may disrupt the body's own histamine degradation, leading to numerous histamine-induced symptoms. Healthy people have two effective enzymatic barriers (please refer to "degradation of histamine").
Histamine in animals and plants
In a little digression, it should be mentioned that histamine is produced and stored in some plants and animals as defense substance. The common nettle for example stores histamine (alongside other substances) in its urticating hairs (trichomes) and injects these upon contact, causing a painful sting. When in danger, the grasshopper, Poekilocerus bufonius (Pyrgomorphidae), secrets a substance, which in addition to cardenolides contains about 1% of histamine. Histamine is also secreted by the cutaneous glands in the skin of some Leptodactylidae frogs. Furthermore, animals and plant antigens such as the mast-cell degranulating peptide (MCD peptide) in the bumblebee venom may release histamine from the mast cells of higher animals and trigger an inflammatory response.
Degradation of histamine
Histamine may be metabolized through four mechanisms, whereby the first two mentioned constitute the primary degradation pathways:
- Oxidative deamination through diamine oxidase (DAO, histaminase).
Chemical equation: Histamine + H2O + O2 => (imidazole-4-yl)acetaldehyde + NH3 + H2O2 - Cyclical methylation through histamine N-methyltransferase (HNMT).
The product resulting from the reaction is N-methylhistamine (NMH). - Acetylation into acetylhistamine.
(This degradation pathway is primarily important in microbial degradation) - Hydroxylase into hydantoin propionic acid.
(Vitamin C could be a cofactor in hydroxylase reactions, which convert histamine into hydantoin propionic acid - analogous to histidine degradation into hydantoin propionate. Presumably, this degradation mechanism is less significant in terms of quantity)
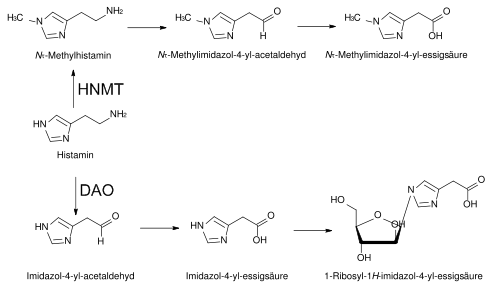
The two primary degradation pathways of histamine [Source: Wikipedia].
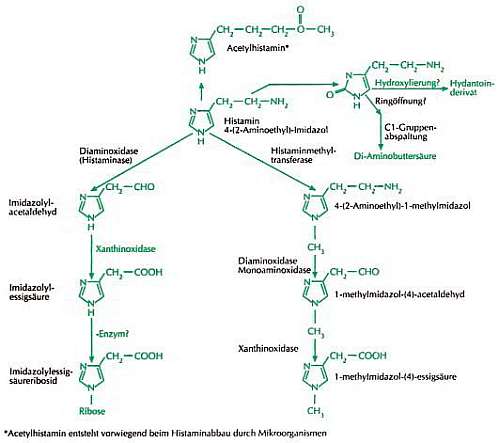
All four known, respectively postulated degradation methods of histamine [Bielenberg 2005].
DAO is excreted by the cells as secretory protein and is responsible for breaking down histamine outside the cells (extracellular), while HNMT only degrades histamine as cytosolic protein within the cells (intracellular), for example in the liver or in the brain.
DAO is primarily synthesized in the cells of the bowel mucosa and excreted into the intestinal lumen, where the DAO mixes with the bolus and breaks down most of the present histamine. This way, DAO creates a first barrier against the entry of histamine into the body.
Histamine, which is absorbed into the body via the intestinal mucosa, reaches the bloodstream and is transported towards the liver. In the liver, the primary detoxifying organ of the body, the degradation takes place with the aid of HNMT.
Histamine released into the cells from vesicles within the body is broken down by HNMT intracellularly.
Guided tour: Go to
histaminosis > causes overview
References and bibliography
The "back"-button of your browser takes you back to the previous position.
| B | Back to the previous position |
|---|---|
| Baenziger et al. 1994 | Baenziger NL, Mack P, Jong YJ, Dalemar LR, Perez N, Lindberg C, Wilhelm B, Haddock RC.: "An environmentally regulated receptor for diamine oxidase modulates human endothelial cell/fibroblast histamine degradative uptake". J Biol Chem. 1994 May 27;269(21):14892-8. https://pubmed.ncbi.nlm.nih.gov/8195119 Die DAO ist ein membrangebundenes Enzym, das an DAO-Rezeptoren auf der Zelloberfläche gebunden wird. Die Rezeptordichte kann in Reaktion auf diverse äussere Einflüsse aktiv reguliert werden. |
| Barnes and Hough 2002 | Barnes WG, Hough LB.: "Membrane-bound histamine N-methyltransferase in mouse brain: possible role in the synaptic inactivation of neuronal histamine.". J Neurochem. 2002 Sep;82(5):1262-71. https://pubmed.ncbi.nlm.nih.gov/12358773 (Im Zentralnervensystem ist Histamin ein Neurotransmitter, der durch die HNMT deaktiviert wird. Die HNMT ist ein lösliches Enzym im Zellplasma. Noch ungeklärt ist, wie das Histamin, das von den Nervenzellen nach aussen in den synaptischen Spalt ausgeschüttet wird, von der im Zellinneren lokalisierten HNMT deaktiviert werden kann. Die (nur in sehr geringem Ausmass stattfindende) Aufnahme von Histamin in die Nervenzelle kann dabei nur eine untergeordnete Rolle spielen. Hingegen konnte gezeigt werden, dass die Membranfraktion der Zellen am Histaminabbau beteiligt sein muss. Man vermutet, dass es eine membranständige HNMT gibt. Deren Existenz konnte noch nicht nachgewiesen werden, doch es muss irgend einen Histamin-Abbaumechanismus ausserhalb der Zelle geben. [Anmerkung: Die DAO kann es nicht sein, da sie im Nervensystem nicht gebildet wird.]) |
| Binda 2002 @ rcsb.org | Image from the RCSB PDB (www.rcsb.org) of PDB ID 1GOS.Binda C, Newton-Vinson P, Hubálek F, Edmondson DE, Mattevi A.: "Structure of human monoamine oxidase B, a drug target for the treatment of neurological disorders.". Nat Struct Biol. 2002 Jan;9(1):22-6. http://www.rcsb.org/pdb/explore.do?structureId=1gos https://pubmed.ncbi.nlm.nih.gov/11753429 (Struktur der menschlichen Monoaminoxidase B (MAO B)) |
| Bielenberg 2005 | Bielenberg, Jens: "Korrelate einer schadstoffinduzierten Veränderung des Histaminstoffwechsels? Die Allergie-Hypothese". Österreichische Apotheker-Zeitung ÖAZ Aktuell (Ausgabe 15/2005), Hauptartikel 15/2005. Anschrift des Autors: Apotheker Jens Bielenberg, Raphael-Apotheke, D-25364 Westerhorn, Bahnhofstr. 53 Der Artikel ist inzwischen online nicht mehr auffindbar. Ein sehr ähnlicher Text desselben Autors ist hier abrufbar: Bielenberg, Jens: "Die Allergie-Hypothese – Fakt oder Fiktion? Allergien - Korrelate einer schadstoffinduzierten Veränderung des Histaminstoffwechsels?". Ärztezeitschrift für Naturheilverfahren 46, 11 (2005).https://www.yumpu.com/de/document/view/6248824/die-allergie-hypothese-fakt-oder-fiktion-zentralverband-der- (Interessanter Artikel über mögliche Ursachen von Allergien, Störfaktoren im Histaminstoffwechsel, Abbaumechanismen von Histamin, Ascorbate (Vitamin C), Umweltschadstoffe, Schilddrüsenerkrankungen) |
| D | Back to the previous position |
| Dahlin et al. 2007 | A. Dahlin, L. Xia, W. Kong, R. Hevner, and J. Wang: "Expression and Immunolocalization of the Plasma Membrane Monoamine Transporter in the Brain". Neuroscience. 2007 May 25; 146(3): 1193–1211. Published online 2007 April 3. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2683847/ |
| Daws 2008 | Lynette C. Daws: "Unfaithful neurotransmitter transporters: Focus on serotonin uptake and implications for antidepressant efficacy". Pharmacol Ther. 2009 January; 121(1): 89–99. Published online 2008 October 29. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2739988/ |
| Duan and Wang 2010 | Haichuan Duan and Joanne Wang: "Selective Transport of Monoamine Neurotransmitters by Human Plasma Membrane Monoamine Transporter and Organic Cation Transporter 3". J Pharmacol Exp Ther. 2010 December; 335(3): 743–753. doi: 10.1124/jpet.110.170142 http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2993547/ |
| F | Back to the previous position |
| Ferk et al. 2012 | Polonca Ferk, Metoda Lipnik-Štangelj, Mojca Kržan, and Katarina Cerne: "Expression of organic cation transporter 3 (SLC22A3) and plasma membrane monoamine transporter (SLC29A4) in human umbilical vein endothelial cells and their relevance for histamine uptake". BMC Pharmacol Toxicol. 2012; 13(Suppl 1): A73. Published online 2012 September 17. doi: 10.1186/2050-6511-13-S1-A73 http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3506340/ |
| G | Back to the previous position |
| Girard et al. 1994 | Girard B, Otterness DM, Wood TC, Honchel R, Wieben ED, Weinshilboum RM.: "Human histamine N-methyltransferase pharmacogenetics: cloning and expression of kidney cDNA". Mol Pharmacol. 1994 Mar;45(3):461-8. https://pubmed.ncbi.nlm.nih.gov/8145732 |
| Geha et al. 2001 | Geha RM, Rebrin I, Chen K, Shih JC: "Substrate and inhibitor specificities for human monoamine oxidase A and B are influenced by a single amino acid". J Biol Chem. 2001 Mar 30;276(13):9877-82. Epub 2000 Dec 29. http://www.jbc.org/content/276/13/9877.long |
| H | Back to the previous position |
| Hough 1999 | Hough, Lindsay B.: "Histamine". American Society for Neurochemistry, 1999. http://www.ncbi.nlm.nih.gov/books/NBK20394/ (Über die Rolle von Histamin im Nervensystem. Zusammenfassende Übersetzung hier) |
| J | Back to the previous position |
| Jarisch 2004 | Jarisch, Reinhart: "Histamin-Intoleranz, Histamin-Intoleranz und Seekrankheit", Thieme-Verlag, 2. Auflage, 2004. ISBN 3-13-105382-8 |
| K | Back to the previous position |
| Kiehl und Ionescu 1989 | Kiehl R, Ionescu G.: "[Histamine degrading enzymes in atopic eczema]. Histamin abbauende Enzyme bei atopischem Ekzem." Z Hautkr. 1989 Dec 15;64(12):1121-3. https://pubmed.ncbi.nlm.nih.gov/2517372 (Increased plasma histamine levels were associated with significantly lowered diamine and type B monoamine oxidase activities in platelet rich plasma of atopic eczema (AE) patients. The diamine oxidase has almost normal cofactor levels (pyridoxal phosphate and Cu2+) but the cofactor levels for type B monoamine oxidase (flavin adenine dinucleotide and Fe2+) are lowered.) |
| Kitanaka et al. 2001 | Kitanaka J, Kitanaka N, Tsujimura T, Kakihana M, Terada N, Takemura M.: "Guinea pig histamine N-methyltransferase: cDNA cloning and mRNA distribution." Jpn J Pharmacol. 2001 Jan;85(1):105-8. https://www.ncbi.nlm.nih.gov/pubmed/11243563 (Verteilung der HNMT im Körper.) |
| L | Back to the previous position |
| Lin et al. 2011 | Zhicheng Lin, Juan J. Canales, Thröstur Björgvinsson, Morgane M. Thomsen, Hong Qu, Qing-Rong Liu, Gonzalo E. Torres, and S. Barak Caine: "Monoamine Transporters: Vulnerable and Vital Doorkeepers". Prog Mol Biol Transl Sci. 2011; 98: 1–46. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3321928/ |
| M | Back to the previous position |
| Maintz et al. 2006 | Maintz, Laura; Bieber, Thomas; Novak, Natalija: "Die verschiedenen Gesichter der Histaminintoleranz: Konsequenzen für die Praxis (Histamine Intolerance in Clinical Practice)", Deutsches Ärzteblatt 2006; 103(51-52). http://www.aerzteblatt.de/archiv/53958, abgerufen am 25.08.2009. |
| Maintz and Novak 2007 | Maintz L, Novak N.: "Histamine and histamine intolerance.", Am J Clin Nutr. 2007 May;85(5):1185-96. Department of Dermatology, University of Bonn, Bonn, Germany. https://pubmed.ncbi.nlm.nih.gov/17490952) |
| Mannaioni et al. 1993 | P. F. Mannaioni, M. G. Di Bello, S. Raspanti, F. Gambassi, L. Mugnai and E. Masini: "Histamine Release from Mast Cells and Basophils. Histamine release by human platelets." Inflammation Research, Volume 38, Numbers 3-4 (1993), C203-C205, DOI: 10.1007/BF01996460. http://www.springerlink.com/content/x0063474j4685x12/) |
| Marshall 1984 | Marshall I.: "Characterization and distribution of histamine H1- and H2-receptors in precapillary vessels." J Cardiovasc Pharmacol. 1984;6 Suppl 4:S587-97. https://www.ncbi.nlm.nih.gov/pubmed/6083401 (Blutgefässe und Histaminrezeptoren.) |
| Masini et al. 1994 | Masini E, Di Bello MG, Raspanti S, Sacchi TB, Maggi E, Mannaioni PF.: "Platelet aggregation and histamine release by immunological stimuli.", Immunopharmacology. 1994 Jul-Aug;28(1):19-29. http://www.ncbi.nlm.nih.gov/pubmed/7523332) |
| McGrath et al. 2009 | McGrath AP, Hilmer KM, Collyer CA, Shepard EM, Elmore BO, Brown DE, Dooley DM, Guss JM.: "Structure and inhibition of human diamine oxidase". Biochemistry. 2009 Oct 20;48(41):9810-22. http://www.ncbi.nlm.nih.gov/pubmed/19764817 (Struktur und Hemmstoffe der DAO.) |
| N | Back to the previous position |
| Nair, Ahmed and Kin 1993 | Nair NP, Ahmed SK, Kin NM.: "Biochemistry and pharmacology of reversible inhibitors of MAO-A agents: focus on moclobemide." J Psychiatry Neurosci. 1993 Nov;18(5):214-25. https://www.ncbi.nlm.nih.gov/pubmed/7905288 ("The enzymes are usually present on the outer mitochondrial membrane of both neuronal and non-neuronal cells (Greenwalt 1972). Most human tissues contain both types of MAO. In the periphery, MAO-A is found mostly in the sympathetic terminals, intestinal mucosa and placenta, whereas MAO-B predominates in the liver, lungs and blood platelets (Da Prada et al 1988; Youdim et al 1980).") |
| O | Back to the previous position |
| Ochiai et al. 2006 | Ochiai Y, Itoh K, Sakurai E, Adachi M, Tanaka Y.: "Substrate selectivity of monoamine oxidase A, monoamine oxidase B, diamine oxidase, and semicarbazide-sensitive amine oxidase in COS-1 expression systems." Biol Pharm Bull. 2006 Dec;29(12):2362-6. https://pubmed.ncbi.nlm.nih.gov/17142964 (Welches Enzym baut welche Amine ab?) |
| Ogasawara et al. 2006 | Ogasawara M, Yamauchi K, Satoh Y, Yamaji R, Inui K, Jonker JW, Schinkel AH, Maeyama K.: "Recent advances in molecular pharmacology of the histamine systems: organic cation transporters as a histamine transporter and histamine metabolism.". J Pharmacol Sci. 2006 May;101(1):24-30. Epub 2006 Apr 28. https://pubmed.ncbi.nlm.nih.gov/16648665 (Die HNMT ist im Inneren der Zellen lokalisiert und deaktiviert Histamin hauptsächlich in Bronchien, Niere und dem Zentralnervensystem. Histamin kann nicht von selbst durch die Zellmembran hindurch ins Zellinnere gelangen. Man vermutete, dass die HNMT trotzdem am Abbau von Histamin, das von ausserhalb kommt, beteiligt sein kann. Dies entweder indem die HNMT auch in die Zellmembran eingebaut sein könnte und auf diese Weise Histamin ausserhalb der Zelle inaktivieren kann, oder indem Transportmechanismen das Histamin aktiv in die Zelle hinein transportieren. Die organischen Kationentransporter OCT2 und OCT3 könnten tatsächlich als Histamintransporter agieren, wie Experimente mit Mäusen zeigten. Die höhere Sterblichkeit der Mäuse mit ausgeschaltetem OCT3 erklärt man sich so, dass der dadurch erhöhte Histaminspiegel die Histaminrezeptoren der Mastzellen, Makrophagen und dendritischen Zellen stimuliert und dadurch die Funktion der Immunzellen verändert haben könnte.) |
| P | Back to the previous position |
| Potera 2012 | Carol Potera: "Common bacterium induces histamine production in neutrophils". Environ Health Perspect. 2012 May;120(5):A190. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3346795/pdf/ehp.120-a190.pdf (Neutrophile Granulozyten (weisse Blutkörperchen) gehören ebenfalls zu den Histamin produzierenden Zelltypen und erhöhen die Histaminproduktion um ein Mehrfaches bei Kontakt mit bestimmten Bakterienarten.) |
| R | Back to the previous position |
| Rozov et al. 2014 | Rozov SV, Zant JC, Karlstedt K, Porkka-Heiskanen T, Panula P.: "Periodic properties of the histaminergic system of the mouse brain." Eur J Neurosci. 2014 Jan;39(2):218-28. doi: 10.1111/ejn.12397. Epub 2013 Nov 6. https://pubmed.ncbi.nlm.nih.gov/24438489 |
| S | Back to the previous position |
| Scinexx 2009 | "Glückshormon reguliert auch Zuckerstoffwechsel. Wirkmechanismus von Serotonin in der Bauchspeicheldrüse aufgeklärt.", Wissensmagazin www.scinexx.de, Springer Verlag. www.scinexx.de/wissen-aktuell-10727-2009-10-29.html, abgerufen am 29.10.2009. Siehe auch folgende Primärliteratur: Paulmann, N. et al.: "Intracellular serotonin modulates insulin secretion from pancreatic ß-cells by protein serotonylation.", PLoS Biol., 7 (2009) e1000229. doi:10.1371/journal.pbio.1000229. Walther, D.J. et al.: "Serotonylation of Small GTPases is a Signal Transduction Pathway that Triggers Platelet alpha-Granule Release", Cell, 115 (2003) 851-862. |
| V | Back to the previous position |
| Vowinckel 2012 | Vowinckel, Jakob: "Die Protein-Monoaminylierung als regulatorischer Mechanismus in der Signaltransduktion; [Protein-monoaminylation - a novel regulatory mechanism in signal transduction]", Dissertation, Freie Universität Berlin. http://www.diss.fu-berlin.de/diss/receive/FUDISS_thesis_000000023285. |
| W | Back to the previous position |
| Walther, Stahlberg und Vowinckel 2011 | Walther DJ, Stahlberg S, Vowinckel J.: "Novel roles for biogenic monoamines: from monoamines in transglutaminase-mediated post-translational protein modification to monoaminylation deregulation diseases.", FEBS J. 2011 Dec;278(24):4740-55. doi: 10.1111/j.1742-4658.2011.08347.x. Epub 2011 Nov 21. https://pubmed.ncbi.nlm.nih.gov/21923757 |
| Walther 2007 | Walther, D.J.: "Protein-Monoaminylierung: Neu erkannte Funktionen monoaminerger Hormone", Jahrbuch 2007, Max-Planck-Institut für molekulare Genetik. www.mpg.de/463476/forschungsSchwerpunkt1 |
| Wikipedia: Histamin | Wikipedia-Artikel: Histamin. de.wikipedia.org/wiki/Histamin, abgerufen am 25.08.2009 |
| X | Back to the previous position |
| Xia et al. 2006 | Li Xia, Karen Engel, Mingyan Zhou, and Joanne Wang: "Membrane localization and pH-dependent transport of a newly cloned organic cation transporter (PMAT) in kidney cells". Am J Physiol Renal Physiol. 2007 February; 292(2): F682–F690. Published online 2006 October 3. doi: 10.1152/ajprenal.00302.2006. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2672957/ |
| Xu et al. 2012 | Xu X, Zhang H, Song Y, Lynch SV, Lowell CA, Wiener-Kronish JP, Caughey GH.: "Strain-dependent induction of neutrophil histamine production and cell death by Pseudomonas aeruginosa.". J Leukoc Biol. 2012 Feb;91(2):275-84. Epub 2011 Nov 10. https://pubmed.ncbi.nlm.nih.gov/22075928 (Neutrophile Granulozyten (weisse Blutkörperchen) gehören ebenfalls zu den Histamin produzierenden Zelltypen und erhöhen die Histaminproduktion um ein Mehrfaches bei Kontakt mit bestimmten Bakterienarten.) |
| Y | Back to the previous position |
| Yoshikawa et al. 2013 | Yoshikawa T, Naganuma F, Iida T, Nakamura T, Harada R, Mohsen AS, Kasajima A, Sasano H, Yanai K.: "Molecular mechanism of histamine clearance by primary human astrocytes". Glia. 2013 Jun;61(6):905-16. doi: 10.1002/glia.22484. Epub 2013 Mar 15. https://pubmed.ncbi.nlm.nih.gov/23505051 |
![[SIGHI]](../pics/logo_sighi_160x160_terracotta.png)